Fig 1 Schematic illustration of the two-step decellularization (TSD) processes of the bovine pericardial bioprosthetic valves. First, the BP was decellularized by Triton X-100, sodium deoxycholate (SDC), DNase I and RNase to get a decellularized bovine pericardium (D-BP), followed by glutaraldehyde (GA) cross-linking. Second, glutaraldehyde-treated bovine pericardium (GA-BP) was post-treated with isopropanol (IPA) and a mild biosurfactant—n-dodecyl-β-D-maltoside (DDM). The final product IPA-DDM-BP or TSD-BP was implanted in rats to assess the extent of in vivo calcification following the ISO standard for corresponding medical implants. The calcification of tissue collagen in the implanted bioprosthetic heart valves (BHVs) was also observed via field emitting SEM. The red spots refer to the residual hazardous substances after the 1st decellularization, mainly some internally phospholipids originally in cell membranes and externally toxic surfactants such as SDC and Triton X-100. These substrates are expected to be replaced by the mild biosurfactants during the post-treatment or the 2nd decellularization.

A Biosurfactant-containing TSD Strategy to Modify Bovine Pericardial Bioprosthetic Valves for Anticalcification
- Chinese Journal of Polymer Science Vol. 41, Issue 1, Pages: 51-66(2023)
DOI:10.1007/s10118-022-2843-9
Scan for full text
Cite this article
Abstract
Bioprosthetic heart valves (BHVs) are important for transcatheter valve replacement. Current commercial BHVs on the market are basically porcine or bovine pericardium (BP) crosslinked with glutaraldehyde (GA). Simply applying GA to BHVs can enhance mechanical stability, but cannot alleviate in vivo calcification. In this work, we developed a two-step decellularization (TSD) strategy to modify this biomacromolecular network, in which BP was post-treated, as the second step of decellularization, with a mild biosurfactant n-dodecyl-β-D-maltoside in a mixture of isopropanol and phosphate-buffered saline after the first step of traditional decellularization and GA cross-linking. The TSD-treated BP exhibited not only low cytotoxicity and excellent mechanical properties in vitro, but also low immune responses and significant anticalcification in vivo. After 60 days of subcutaneous implantation in the back of Wistar rats, the calcium content was, as quantified with an inductively coupled plasma optical emission spectrometer, only 1.1 μg/mg compared to 138.6 μg/mg in the control group without the post-treatment. In addition, collagen fibrils were observed with field emitting scanning electron microscopy (SEM), and the morphology and composition of the calcified sites resulting from in vivo biomineralization were studied with SEM with energy dispersive spectroscopy and also X-ray diffraction. This study proposes a facile yet effective anticalcification strategy for the modification of the bovine pericardial bioprosthetic heart valve, a natural biomacromolecular network.
Keywords
Heart valve;
Anticalcification;
Collagen;
In vivo biomineralization ;
Transcatheter aortic valve implantation (TAVI);
Extracellular matrix;
Biomacromolecular network;
Biosurfactant;
Bovine pericardium
INTRODUCTION
The heart valve maintains human blood circulation by achieving unidirectional blood flow. Valvular heart disease is caused by valve stenosis and regurgitation, which is a leading cause of morbidity and mortality.[
The commercial BHVs in clinics are basically porcine pericardium (PP) or bovine pericardium (BP) treated by glutaraldehyde (GA). GA can significantly improve the mechanical property of a collagen network and the resistance to enzymatic hydrolysis owing to its excellent cross-linking ability.[
Thereinto, calcification is the major cause of bioprosthetic valve failure.[
Herein, we developed a two-step decellularization (TSD) strategy, in which the first step of traditional decellularization and GA cross-linking was followed by the second step of treatment of n-dodecyl-β-D-maltoside (DDM) in the mixture of isopropanol (IPA) and phosphate-buffered saline (PBS). The basic procedures are schematically presented in
EXPERIMENTAL
Materials
Fresh BP was provided by Lifetech Scientific Co. Ltd. (Shenzhen, China). GA (25 wt%), IPA, DDM and any other surfactants were purchased from Aladdin (Shanghai, China). Collagenase II was a product of Sigma-Aldrich (US). PBS (pH 7.4), fetal bovine serum (FBS), Tris-HCl, penicillin-streptomycin-amphotericin B, 4% paraformaldehyde (PFA) solution, 4,6-diamidino-2-phenylindole (DAPI), phalloidin-rhodamine solution and any other cell culture medium were purchased from Beyotime Biotechnology (Shanghai, China). Human umbilical vein endothelial cells (HUVECs) were purchased from Shanghai Institute of Biochemistry and Cell Biology (Shanghai, China). Cell counting kit-8 (CCK-8) was from Dojondo (Japan). Alizarin red, Masson, hematoxylin-eosin staining (HE) and all antibody reagents were purchased from Servicebio (Shanghai, China).
Fabrication of BP Bioprosthetic Valves
In order to fabricate decellularized bovine pericardium (D-BP), the fresh BP collected from the slaughterhouse was immediately washed with 4 °C normal saline within 4 h, and then immersed in a mixed solution containing 0.5 wt% Triton X-100 and 0.5 wt% SDC for 12 h. After being rinsed with PBS, the specimens were treated with 50 U/mL DNase I and 1000 μg/mL RNase overnight at 37 °C. The enzyme dilution was Tris buffer (10 mmol/L Tris-HCl at pH 7.5, 2.5 mmol/L MgCl2, 0.1 mmol/L CaCl2). Finally, the specimens were rinsed repeatedly with normal saline to thoroughly wash off the surface activity and enzyme residues on the BP. D-BP was stored in PBS containing penicillin (100 U/mL), streptomycin (0.1 mg/mL), and amphotericin B (0.25 μg/mL) at 4 °C.
As the fabrication of GA-BP was concerned, the above D-BP was spread out on a fixed plate to prevent curling and then placed in a 0.625 wt% GA solution (pH 7.4) at room temperature (25 °C) to experience cross-linking for 72 h. GA-BP was thus obtained.
In terms of fabrication of IPA-DDM-BP, namely TSD-BP, the GA-BP was cut into small pieces of about 10 cm × 10 cm, and post-treated in a GA solution (pH 7.4) containing 20 wt% IPA, and 0.5 wt% DDM at 37 °C for 12 h. The reagent residues were repeatedly washed out with PBS. The obtained modified valve IPA-DDM-BP or TSD-BP was stored in 0.625 wt% GA solution (pH 7.4).
Physicochemical Characterizations of BPs before and after Treatments
Prior to morphology observation, specimens of D-BP, GA-BP, and TSD-BP were freeze-dried and cut into a size of 50 mm × 50 mm. Gold was sprayed under 10 mA for 90 s. The morphology of collagen fibrils was observed at an accelerating voltage of 3 kV with a scanning electron microscope (SEM, Ultra 55, Germany).
The cross-linking properties of BP valves were characterized in a thermal shrinkage thermometer (JY-PS-83, Yuanmore Co., Ltd, China). The BP tissue specimens of each group were cut into standard long splines of 56 mm × 3 mm (n=5), and the two ends of the splines were hung on the two hooks of the heat shrinkage apparatus. The specimens were then immersed in normal saline to heat from 20 °C to 90 °C at a rate of 2 °C/min. The test method refers to the standard QB/T 2713—Leather Physical and Mechanical Test Determination of Shrinkage Temperature.
Differential scanning calorimetry (DSC, TA Q2000, USA) is a usual tool to measure the thermal denaturation temperature of a biomacromolecule-containing material. The specimens were thoroughly rinsed with PBS and then freeze-dried. The dried specimens were weighed (10 mg per specimen) and sealed in aluminium crucibles. The test temperature ranged from 30 °C to 140 °C at a heating rate of 10 °C/min.
The contact angle test was used to characterize the hydrophilicity of the sample surface, and the static water contact angle (WCA) was measured with a contact angle meter (JC2000DM, Zhongchen). Briefly, 10 μL of deionized water was dripped on the surface of the specimen and a camera was used to take a picture. Contact angles were viewed on the monitor's screen by using SCA 20 software. Four parallel samples were taken for each specimen.
In order to test tensile performance, D-BP, GA-BP and TSD-BP were uniformly cut into dumbbell shapes of 57.5 mm × 12.5 mm (n=7 for each group) along the fiber direction with a standard dumbbell steel die. Specimens stored in normal saline were slightly dried with paper and then tested with a universal tensile testing machine (Instron 5966, USA) using a 100 N sensor at a tensile rate of 50 mm/min. The stress-strain curve of the specimen was recorded, and the elastic modulus and the breaking stress of the rope were measured. Data for specimens with fractures in the grips were excluded from the statistical analysis.
Collagen stability in fresh and cross-linked BPs was examined by collagenase treatment. The lyophilized D-BP, GA-BP and TSD-BP were cut into 1 cm × 1 cm (n=5 for each group). The initial weight of each specimen was recorded as W0. The dried valve was then soaked in PBS (pH 7.4) containing collagenase II (125 U/mL, pH 7.4) and placed in a constant temperature shaker at 37 °C with 60 r/min for 24 h. The specimen was washed with PBS and lyophilized again. After the moisture was completely removed, a specimen was weighed as Wt. The weight loss rate upon enzymatic degradation is ready to be calculated from
CollagenHydrolysis=W0−WtW0×100% 1
Characterization of Cytocompatibility
We designed two protocols for evaluating the difference of toxicity of surfactants. Experiment 1: after different groups of surfactants including sodium dodecyl sulfate (SDS), sodium deoxycholate (SDC), Triton X-100, Tween 80 and DDM were incubated in a 48-well plate for 24 h (n=4), every well was washed twice with PBS solution soaking for 5 min each time and discarded. Here we took the PBS solution as the blank control. After culturing HUVECs (EA.hy926) for 1 day and 3 days, we accessed cytotoxicity using CCK-8. Experiment 2: This experiment is to further explore the cytotoxicity followed by washing with DDM after incubation with different groups of surfactants. After different groups of surfactants were incubated in a 48-well plate for 24 h (n=4), the original surfactant solution was discarded, and 0.5% DDM was added to incubate for 24 h more. Then, every well was washed twice with PBS solution (soaking for 5 min each time) and discarded. Lastly, we performed cell seeding and cell viability test similar to Experiment 1.
The cytotoxicity of BP was evaluated according to ISO 10993-5:2009/GB/T 16886.5—Biological Evaluation of Medical Devices Part 5. An extraction method was used in this experiment. GA-BP and TSD-BP specimens were cut into 1 cm × 1 cm (n=4), placed in a 24-well plate, soaked in 75% ethanol solution for sterilization and washed with PBS. Subsequently, 1 mL of high-glucose DMEM containing 10% FBS, 1% penicillin-streptomycin, and 1% glutamine was added to continue the culture. The samples were incubated in a sterile CO2 incubator at 37 °C for 72 h to obtain the material extract. HUVECs (EA.hy926) was chosen to study the cytotoxicity of the samples. Cells were seeded in 96-well cell culture plates at a density of 1×104 cells per well. After culturing for 24 h, the original medium was discarded and replaced with the sample extract, and incubated for another 24 or 72 h. Add 100 μL of 10% CCK-8 solution to the well and incubate at 37 °C for 1 h. After discarding the leaching solution, 100 μL of 10% CCK-8 solution was added to each well and incubated at 37 °C for 1 h. Finally, the absorbance of each specimen at 450 nm was measured with a multifunctional microporous plate detector (BioTek, Cytation3, USA). The medium without any sample was used as the blank control of cytotoxicity.
In order to carry out endothelial cell adhesion tests, GA-BP and TSD-BP were cut into 1cm × 1cm (n=4) and placed in a 24-well plate. The specimens were soaked in a 75% ethanol solution for sterilization and washed with PBS. After that, human endothelial cells were seeded on the sample surface at a density of 1×104 cells per well. The culture lasted for 24 h. At room temperature, the cells were stained with 300 μL of 1 μg/mL phalloidin-rhodamine staining solution in the dark for 30 min, and 2 μg/mL of DAPI staining solution in the dark for 8 min. After staining, the adhesion of endothelial cells to the sample surface was observed with a fluorescence inverted microscope (Axio Scope A1, Carl Zeiss Optics, Germany).
Characterization of Hemocompatibility
Prior to platelet adsorption tests, the whole blood of a rabbit was collected using sodium citrate anticoagulant tubes. The blood was centrifuged at 100 g for 10 min, and the supernatant was centrifuged again at 400 g for 10 min to take the transition layer to obtain platelet-rich plasma (PRP). The specimens of GA-BP (control) and TSD-BP (φ=10 mm) were placed in a 24-well plate, wetted with PBS, which was then discarded. Then, 1 mL of PRP was added and incubated at 37 °C for 1 h. Discard the PRP and rinse the sample for 3 times with PBS. Afterwards the specimens were fixed with 4% paraformaldehyde for 10 min and then rinsed for 3 times with PBS. After washed and dehydrated with a series of gradients of alcohol solutions (30%, 50%, 75%, 90%, and 100%, each for 20 min), the specimens were dried at room temperature, sprayed with gold at 10 mA for 90 s. The images of platelet adsorption were captured at an accelerating voltage of 3 kV with a scanning electron microscope (SEM, Ultra 55, Germany).
In order to carry out hemolysis tests, we took 8 mL of fresh rabbit anticoagulant and diluted it with 10 mL of 0.9% NaCl solution. We cut GA-BP and TSD-BP into 2 cm × 2 cm, put them into a 15 mL centrifuge tube, and added 5 mL of normal saline. After incubated at 37 °C for 30 min, each tube was added with 0.1 mL of the diluted anticoagulant and incubated for 60 min more at 37 °C. Next, we carried out centrifugation at 800 g for 5 min and took the supernatant to measure the absorbance at 540 nm with a multifunctional microplate detector (BioTek, Cytation3, USA). The hemolysis was calculated from the absorbance values. Distilled water and normal saline were used as positive and negative controls, respectively.
Animal Experiments
Specimens were cut into 1 cm × 1 cm and washed thoroughly with sterile PBS solution 3 times before implantation. We selected 3-week-old SPF male Wistar rats with a weight of 45−55 g as our experimental animals. All animal usage obeyed the regulations as set forth in the Animal Welfare Act. Rats were first anesthetized, shaved, and routinely sterilized. A surgical opening of about 1.5 cm was made on the back, and there were totally four pockets on the left and right sides. One specimen was placed in each pocket, and 10 specimens were implanted in each group. Rats were euthanized by cervical dislocation and 5 implants were removed after 30 and 60 days. Half of the explanted tissue was preserved for histology tests; the other half was frozen for calcium analysis.
Prior to in vivo calcification evaluation, we removed the intertwined fibrous capsules and tissues from the sample, washed them with normal saline and dried at 60 °C for 24 h (n=5 for each group). Then we weighed specimens and digested them in 6 N HCl at 97 °C for 24 h. Calcium content was determined using an inductively coupled plasma optical emission spectrometer (ICP-OES, iCAP 7400, Thermo Fisher, US). The morphologies of collagen fibrils and calcified sites were observed at an accelerating voltage of 3 kV with a scanning electron microscope (SEM, Ultra 55, Germany). The elemental mapping of specimens was tested using SEM with energy dispersive spectroscopy (EDS-SEM) (n=4 for each group). The composition of the calcified sites was also characterized by X-ray diffraction (XRD, Bruker D8 diffractometer, Germany) in the range of 10°−70° and Fourier-transform infrared spectroscopy (FTIR).
In order to carry out in vivo immunohistochemical and histological evaluation, another part of the sample was fixed in 4% paraformaldehyde, embedded in paraffin and sectioned. The slices were stained with alizarin red, Masson, hematoxylin and eosin (HE). The HE staining was used for histopathological observation, Masson staining for the spatial distribution of collagen and elastin biomacromolecules, and alizarin red staining for visualization of mineralized calcium salts. Slice scanning was performed with Olympus OlyVIA VS200. In addition, we carried out immunohistochemical staining with cluster of differentiation 3 (CD3), F4/80, tumor necrosis factor alpha (TNF-α) and interleukin 10 (IL-10) markers to analyze inflammation and immune responses. The antibodies used for immunohistochemical detection are listed in Table S1 (in the electronic supplementary information, ESI).
Ethics Statement: in this study, all animal experiments were conducted following the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. We try our best to minimize the suffering of animals.
Statistical Analysis
All experimental results in this study are presented as mean ± standard deviation (SD). Statistical analysis was performed with Origin 2021 (Origin lab Inc., USA). One-way ANOVA was performed to assess significant differences. In this study, “*” stands for p<0.05; “**” stands forp<0.01; and “***” stands forp<0.001; “****” stands for extremely significant difference withp<0.0001.
RESULTS
Decellularization and Characterization of TSD-BP
The extent of decellularization is a key indicator to a bioderived material. If a pericardium is not completely decellularized, residual cells or cellular debris lead to severe immunogenicity and calcification after the valve is implanted.[
Fig 2 Some basic characterizations of BP valves. (a) Optical micrographs of HE-stained histopathological slices to demonstrate the decellularization efficacy; (b) Water contact angles of GA-BP and TSD-BP; (c) SEM images of the indicated specimens.
As shown in Fig. S1 (in ESI), we compared the effects of five decellularization methods. Different detergents led to distinct levels of decellularization. In the single detergent group, strip-shaped (vessel) or dot-shaped (single cell) cell residues were seen in the extracellular matrix (ECM), and the removal of cells was highly insufficient compared with that in the detergent-enzyme group. In addition, ECM was damaged to some extent in the group added with 0.5% SDS. In contrast, the detergent-enzyme group resulted in successful decellularization and preserved the ECM structure. As the water contact angle is concerned, no significant difference was found between the two groups (
Mechanical Properties and Structural Stability
Mechanical properties and structural stability are important for a valve device. Both denaturation temperature and thermal shrinkage temperature reflect cross-linking performance: the higher the temperature, the better the cross-linking.[
Fig 3 Thermodynamic stability of BP valves experiencing different treatments. (a) Typical DSC thermodynamic test curve and the denaturation temperature of the D-BP, GA-BP and TSD-BP; (b) The schematic diagram and statistical results of the thermal shrinkage temperature test up to 100 °C according to QB/T 2713-2005 and ISO 3380:2002: Leather-Physical and Mechanical Tests—Determination of Shrinkage Temperature. The temperature when the BP valve starts to change from flat to shrinking and curling during the heating process is defined as thermal shrinkage temperature. “***” Means a very significant difference with p < 0.001.
Thermal shrinkage temperature test can reflect the crosslinking extent of a BP valve. As depicted in
The mechanical properties of valve materials were further quantified. Typical stress-strain curves in tensile tests are shown in
Fig 4 Mechanical properties and structural stability of the BP valves. (a) Typical stress-strain curve and statistical results of elastic modulus and tensile strength of BP valves; (b) The schematic diagram of triple-helix structure of a collagen biomacromolecule, crosslinking of collagens by GA, collagen hydrolysis by enzymes, and the experimental results of the indicated BP valves experiencing collagenase degradation for 24 h. “***” Means a very significant difference with p<0.001.
The GA cross-linking is aimed not only to improve the mechanical properties, but also to enhance biochemical stability to resist enzymatic hydrolysis and chemical dissociation after the valve is implanted into human body. Therefore, it is necessary to perform an enzymatic hydrolysis test to assess the stability of a valve tissue. As illustrated in
The experimental results from SEM observations, mechanical, thermal stability and anti-enzymolysis tests were consistent with each other, and the GA crosslinking of collagens could improve the heat resistance, enzymatic hydrolysis resistance and mechanical ability of the valve. The post-treatment using biosurfactants did not affect the GA cross-linking performance of the decellularized valves, and the resultant TSD-BPs exhibited excellent mechanical properties, thermodynamic stability and biological stability.
Cytocompatibility of the Modified Valve Materials
Cytotoxicity test is necessary to access the possible harmful effect of a chemical relevant to a medical device or pharmaceutics. According to ISO 10993-5: 2009, the cytotoxicity of a material can be evaluated semi-quantitatively. The reactivity of cells in a leaching solution or in direct contact with the device for 24−72 h was scored on a scale: 0, none; 1, slight; 2, mild; 3, moderate; 4, severe. A biomaterial is considered of acceptable cytotoxicity with Grade 0 or 1. In this study, we used the CCK-8 method of the material leaching solution to test the cytotoxicity. The acceptance criteria for cell viability are 70% according to ISO 10993-5:2009.
Generally speaking, surfactants follow the order of toxicity as cationic surfactants>anionic surfactants>nonionic surfactants.[
Fig 5 Cytocompatibility of the chemicals used for the treatment of BPs and the resultant BP valves. (a) Cell viabilities in the media containing the indicated different surfactants and cytotoxicity tests of the different surfactants after DDM rinsing for 24 h (n=4). The dashed line represents the acceptance criteria for cell viability according to ISO 10993-5:2009. (b) In vitro cytotoxicity evaluation of extracts of the GA-BP (control) and TSD-BP (n=4). (c) Fluorescence micrographs of HUVECs on GA-BP and TSD-BP after incubating for 24 h. The upper and the lower are, respectively, grayscale photographs and color photographs under fluorescence excitation with phalloidin-rhodamine-marked F-actin in red and DAPI-marked nuclei in blue. “***” Means a very significant difference with p<0.001.
We further designed experiments followed by washing with DDM after incubation with different surfactants, which simulates the post-treatment or the second decellularization step in our TSD treatment of the valve. As a consequence, the toxicities of all surfactants were significantly reduced after the DDM treatment. The cell viabilities of the basic culture for 1 day were levelled to 80%, and those of the culture for 3 days were over 96%. These results imply that all the residues have been replaced during the second step of the biosurfactant-containing TSD strategy, namely, the DDM treatment.
We also checked cytotoxicity of the extract medium. As shown in
Both GA-BP and TSD-BP were stored in a 0.625% GA solution, and the valves were washed under the same condition prior to cytotoxicity tests. So, the lower cell viability of the group of GA-BP than that of TSD-BP in
Hemocompatibility
To assess the hemocompatibility of the valve, the platelet adsorption assay was used to examine the coagulation properties of the specimen. TSD-BP showed slightly less adsorption to platelets than that of GA-BP (
Fig 6 Hemocompatibility of BP valves. (a) SEM images of BP surfaces with platelet adsorption; (b) Global views of hemolysis tests and quantitative results. Distilled water and normal saline were used as positive and negative controls, respectively. For each group, n=5. The dashed line represents 5%, the accepted lowest safety standards of hemolysis for blood-contacting materials according to ISO 10993-4:2002.
In vivo Calcification Evaluation
Anticalcification is our primary goal of the modification of bioprosthetic valves. We examined in vivo calcification after implantation the BP sample in Wistar rats. As is schematically shown in
Fig 7 In vivo calcification evaluation of GA-BP, IPA-BP, and IPA-DDM-BP (TSD-BP). (a) Schematic representation of the site of subcutaneous implantation in a Wistar rat and the process for testing calcium content of the implants; (b) The global view of BP valves (1 cm × 1 cm) after being taken out from experimental rats on the indicated days of post-implantation and then dried; (c) Quantitative analysis of calcium content determined via ICP-OES.
We quantified the extent of calcium using ICP-OES, and the results are shown in
We also observed and semi-quantified the element distribution on the dried valves. In terms of calcium content, the EDS-SEM results in
Fig 8 Characterization of calcified specimens 60 days after implantation. (a) The upper images show the elemental mapping of GA-BP and TSD-BP by using scanning electron microscopy with energy dispersive spectroscopy (EDS-SEM). The lower images are typical EDS spectra and quantitative statistics of weight fractions of P and Ca elements of the specimens. (b) Typical XRD spectra of GA-BP and TSD-BP. (c) A typical FTIR spectrum of a calcified specimen.
To further confirm the calcified compounds of GA-BP samples experiencing in vivo biomineralization, we carried out XRD and FTIR measurements. From the XRD spectrum in
Observations of Spatial Distribution and Orientation of Regenerated Collagen Fibrils and Calcified Aggregates in Tissues
The BP is composed of wavy type I collagen fibers and long elastic fibers, which consitutue the basic skeleton of a variety of connective tissues. One collagen molecule is made up of three polypeptide chains forming a superhelix approximately 1.5 nm in diameter.[
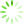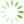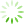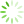
Fig 9 SEM images of in vivo formed calcified sites with both calcium nodes and collagen fibrils. (a) A whole image with magnification ×10000; (b) local images with magnification ×20000 with cobblestone-like (upper row) and coral-like (lower row) morphology.
The microscopic morphology of the attached calcium clusters or nodules was observed in a higher magnification. We classified calcified crystal points roughlly into two types of microscopic morphologies, cobblestone-type and coral-type (
The physiology of connective tissues is highly dependent upon the organization of structural biomacromolecules, in particular, collagen.[
Fig 10 Optical micrographs of tissue slides experiencing the indicated stainings. The slicing was made after taking out the valves from rats after subcutaneous implantation for 60 days.
In vivo Immunization Evaluation
To explore the immune response of the external biomaterials, TSD-BP (experimental group) and GA-BP (control group) implanted in rats subcutaneously for 30 and 60 days were taken out for immunohistochemical staining. T cells are key cells of the adaptive immune system, and the characteristic marker of T cells is the cluster of differentiation 3 (CD3), a membrane protein. Macrophages belong to a core innate immune cell type; they functionally secrete a wide variety of cytokines and chemokines, which makes these cells powerful regulators in the host immune system. F4/80 is a marker for mature murine macrophages, which is divided into two types according to the stimulation-induced polarization, M1 and M2. M1 macrophages are pro-inflammatory and secrete inflammatory cytokines and chemokines, such as tumor necrosis factor-α (TNF-α). M2 macrophages are thought to have immunosuppressive effects and secrete anti-inflammatory factors such as interleukin 10 (IL-10), which affect tissue remodelling process of the valve in vivo.[
Fig 11 Immune responses induced by GA-BP (control group) and TSD-BP (experimental group) after subcutaneous implantation in rats for 30 and 60 days. (a) Optical micrographs. Immunohistochemical staining was performed for sections towards CD3, F4/80, TNF-α and IL-10 markers. (b) Quantitative statistics of positive cells. For each specimen, three sites were randomly selected and Image J software was used for data collection. 100% means that all cells within the statistical field of view are antibody immunopositive. “**” and “***” mean significant differences with p < 0.01 and p < 0.001, respectively.
DISCUSSION
Structural biomacromolecules are complementary to synthetic polymers,[
Calcification is related to collagen-relevant mechanical injury in BPs, as revealed by an in vitro calcification experiment on valves after uniaxial cyclic stretching of 1 million times or failure.[
Calcification is a serious problem limiting the lifespan of a bioprosthetic valve in clinical applications. Phospholipids can bind calcium and play an essential role in the formation of calcium phosphate crystals. Combination of alcohol and surfactants has been reported to reduce calcification. In 1989, Jones et al. first used surfactants as an anticalcification treatment for bioprosthetic valves; they evaluated the impacts of four kinds of reagents (surfactants, phosphoramidate, toluidine blue and polyacrylamide) to valvular tissues for the mitral and tricuspid replacement in sheep and found that only the surfactants substantially reduced calcification.[
In this study, we introduced n-dodecyl-β-D-maltoside (DDM) into the TSD strategy for the first time. Its chemical structure is shown in
Our research used a natural plant-derived nonionic surfactant DDM in the post-treatment of GA-fixed valves, instead of any chemically synthesized or semi-synthetic surfactant. The experiment of subcutaneous implantation in Wistar rats proved its effectiveness in anticalcification (
CONCLUSIONS
In this study, we propose a “two-step decellularization” (TSD) strategy by using a biosurfactant at the second stage for the treatment of bioprosthetic valves. Experiments in vitro and in vivo confirmed that BHVs treated by TSD (IPA-DDM treatment following GA crosslinking of the decellularized matrix) exhibited high anticalcification efficacy as well as low cytotoxicity and excellent mechanical properties. In particular, after 60 days of implanting a BHV into a rat, the calcium content in the TSD-treated implant was only 1.1 μg/mg in contrast to 138.6 μg/mg without the biosurfactant-containing post-treatment. In addition, the morphology and composition of the calcified sites were also investigated. The revealed structure of the calcification site with both collagen fibrils and calcified clusters might be helpful for understanding the in vivo biomineralization containing both biomacromolecules and inorganic compounds under tissue microenvironment. This work provides a facile and effective approach to enhance the anticalcification of BHVs. Moreover, the employment of the mild biosurfactant DDM will broaden the horizons for researchers who attempt to improve the service life and safety of interventional bioprosthetic products in the fields of cardiovascular and other biomedical materials.
References
Arsalan, M.; Walther, T . Durability of prostheses for transcatheter aortic valve implantation . Nat. Rev. Cardiol. , 2016 . 13 360 -367 . DOI:10.1038/nrcardio.2016.43 .
Saleeb, S. F.; Gauvreau, K.; Mayer, J. E.; Newburger, J. W . Aortic valve replacement with bovine pericardial tissue valve in children and young adults . Circulation , 2019 . 139 983 -985 . DOI:10.1161/CIRCULATIONAHA.118.037187 .
Kuang, D.; Lei, Y.; Yang, L.; Wang, Y . Preclinical study of a self-expanding pulmonary valve for the treatment of pulmonary valve disease . Regen. Biomater. , 2020 . 7 609 -618 . DOI:10.1093/rb/rbaa035 .
Li, N.; Peng, J.; Tan, M.; Bai, Y.; Qiao, F.; Han, Q.; Lu, F.; Li, B.; Han, L.; Xu, Z.; Zhang, G . Clinical outcome of reoperation for mechanical prosthesis at aortic position . Heart Lung Circ. , 2021 . 30 1084 -1090 . DOI:10.1016/j.hlc.2021.01.002 .
Yacoub, M. H . Establishing pediatric cardiovascular services in the developing world: a wake-up call . Circulation , 2007 . 116 1876 -1878 . DOI:10.1161/CIRCULATIONAHA.107.726265 .
Zilla, P.; Brink, J.; Human, P.; Bezuidenhout, D . Prosthetic heart valves: catering for the few . Biomaterials , 2008 . 29 385 -406 . DOI:10.1016/j.biomaterials.2007.09.033 .
Puri, R.; Chamandi, C.; Rodriguez-Gabella, T.; Rodes-Cabau, J . Future of transcatheter aortic valve implantation-evolving clinical indications . Nat. Rev. Cardiol. , 2018 . 15 57 -65 . DOI:10.1038/nrcardio.2017.116 .
Ma, B.; Wang, X.; Wu, C.; Chang, J . Crosslinking strategies for preparation of extracellular matrix-derived cardiovascular scaffolds . Regen. Biomater. , 2014 . 1 81 -89 . DOI:10.1093/rb/rbu009 .
Fioretta, E. S.; Motta, S. E.; Lintas, V.; Loerakker, S.; Parker, K. K.; Baaijens, F. P. T.; Falk, V.; Hoerstrup, S. P.; Emmert, M. Y . Next-generation tissue-engineered heart valves with repair, remodelling and regeneration capacity . Nat. Rev. Cardiol. , 2021 . 18 92 -116 . DOI:10.1038/s41569-020-0422-8 .
Rémi, E.; Khelil, N.; Di Centa, I.; Roques, C.; Ba, M.; Medjahed-Hamidi, F.; Chaubet, F.; Letourneur, D.; Lansac, E.; Meddahi-Pelle, A. . Pericardial processing: challenges, outcomes and future prospects . Biomater. Sci. Eng. , 2011 . 22 438 -456 . DOI:10.5772/24949 .
Liu, C.; Qiao, W.; Cao, H.; Dai, J.; Li, F.; Shi, J.; Dong, N . A riboflavin-ultraviolet light A-crosslinked decellularized heart valve for improved biomechanical properties, stability, and biocompatibility . Biomater. Sci. , 2020 . 8 2549 -2563 . DOI:10.1039/c9bm01956a .
Fiddler, G. I.; Gerlis, L. M.; Walker, D. R.; Scott, O.; Williams, G. J . Calcification of glutaraldehyde-preserved porcine and bovine xenograft valv& in young children . Ann. Thorac. Surg. , 1983 . 35 257 -261 . DOI:10.1016/S0003-4975(10)61554-8 .
Schinke, T.; McKee, M.; Karsenty, G . Extracellular matrix calcification: where is the action? . Nat. Genet. , 1999 . 21 150 -151 . DOI:10.1038/5928 .
Kim, K. M . Cells, rather than extracellular matrix, nucleate apatite in glutaraldehyde-treated vascular tissue . J. Biomed. Mater. Res. , 2002 . 59 639 -645 . DOI:10.1002/jbm.10038 .
Schoen, F. J.; Levy, R. J . Calcification of tissue heart valve substitutes: progress toward understanding and prevention . Ann. Thorac. Surg. , 2005 . 79 1072 -1080 . DOI:10.1016/j.athoracsur.2004.06.033 .
D'Alessandro, C. C.; Komninou, M. A.; Badria, A . F.; Korossis, S.; Koutsoukos, P.; Mavrilas, D. Calcification assessment of bioprosthetic heart valve tissues using an improved in vitro model . IEEE Trans. Biomed. Eng. , 2020 . 67 2453 -2461 . DOI:10.1109/TBME.2019.2963043 .
Liu, J.; Li, B.; Jing, H.; Qin, Y.; Wu, Y.; Kong, D.; Leng, X.; Wang, Z . Curcumin-crosslinked acellular bovine pericardium for the application of calcification inhibition heart valves . Biomed. Mater. , 2020 . 15 045002 DOI:10.1088/1748-605X/ab6f46 .
Wang, X.; Zhai, W.; Wu, C.; Ma, B.; Zhang, J.; Zhang, H.; Zhu, Z.; Chang, J . Procyanidins-crosslinked aortic elastin scaffolds with distinctive anti-calcification and biological properties . Acta Biomater. , 2015 . 16 81 -93 . DOI:10.1016/j.actbio.2015.01.028 .
Yang, L.; Xie, S.; Ding, K.; Lei, Y.; Wang, Y . The study of dry biological valve crosslinked with a combination of carbodiimide and polyphenol . Regen. Biomater. , 2021 . 8 rbaa049 DOI:10.1093/rb/rbaa049 .
Liang, H. C.; Chang, Y.; Hsu, C. K.; Lee, M. H.; Sung, H. W . Effects of crosslinking degree of an acellular biological tissue on its tissue regeneration pattern . Biomaterials , 2004 . 25 3541 -3552 . DOI:10.1016/j.biomaterials.2003.09.109 .
Hu, Y.; Su, X.; Lei, Y.; Wang, Y . A novel anti-calcification method for bioprosthetic heart valves using dopamine-modified alginate . Polym. Bull. , 2018 . 76 1423 -1434 . DOI:10.1007/s00289-018-2450-7 .
Nogueira, G. M.; Rodas, A. C. D.; Weska, R. F.; Aimoli, C. G.; Higa, O. Z.; Maizato, M.; Leiner, A. A.; Pitombo, R. N. M.; Polakiewicz, B.; Beppu, M. M . Bovine pericardium coated with biopolymeric films as an alternative to prevent calcification: in vitro calcification and cytotoxicity results . Mater. Sci. Eng. C , 2010 . 30 575 -582 . DOI:10.1016/j.msec.2010.02.011 .
Ohri, R.; Hahn, S. K.; Hoffman, A. S.; Stayton, P. S.; Giachelli, C. M . Hyaluronic acid grafting mitigates calcification of glutaraldehyde-fixed bovine pericardium . J. Biomed. Mater. Res. A , 2004 . 70 328 -334 . DOI:10.1002/jbm.a.30088 .
Lei, Y.; Deng, L.; Tang, Y.; Ning, Q.; Lan, X.; Wang, Y . Hybrid pericardium with VEGF-loaded hyaluronic acid hydrogel coating to improve the biological properties of bioprosthetic heart valves . Macromol. Biosci. , 2019 . 19 e1800390 DOI:10.1002/mabi.201800390 .
Luo, Y.; Huang, S.; Ma, L . Zwitterionic hydrogel-coated heart valves with improved endothelialization and anti-calcification properties . Mater. Sci. Eng. C Mater. Biol. Appl. , 2021 . 128 112329 DOI:10.1016/j.msec.2021.112329 .
Vasudev, S. C.; Chandy, T . Polyethylene glycol-grafted bovine pericardium: a novel hybrid tissue resistant to calcification . J. Mater. Sci. Mater. Med. , 1999 . 10 121 -128. .
Kristensen, E . M.; Larsson, R.; Sanchez, J.; Rensmo, H.; Gelius, U.; Siegbahn, H. Heparin coating durability on artificial heart valves studied by XPS and antithrombin binding capacity . Colloids Surf. B Biointerfaces , 2006 . 49 1 -7 . DOI:10.1016/j.colsurfb.2006.02.007 .
Lim, H. G.; Kim, S. H.; Choi, S. Y.; Kim, Y. J . Anticalcification effects of decellularization, solvent, and detoxification treatment for genipin and glutaraldehyde fixation of bovine pericardium . Eur. J. Cardiothorac. Surg. , 2012 . 41 383 -390 . DOI:10.1016/j.ejcts.2011.05.016 .
Tam, H.; Zhang, W.; Feaver, K. R.; Parchment, N.; Sacks, M. S.; Vyavahare, N . A novel crosslinking method for improved tear resistance and biocompatibility of tissue based biomaterials . Biomaterials , 2015 . 66 83 -91 . DOI:10.1016/j.biomaterials.2015.07.011 .
Shen, M.; Kara-Mostefa, A.; Chen, L.; Daudon, M.; Thevenin, M.; Lacour, B.; Carpentier, A . Effect of ethanol and ether in the prevention of calcification of bioprostheses . Ann. Thorac. Surg. , 2001 . 71 S413 -S416 . DOI:10.1016/S0003-4975(01)02521-8 .
Pathak, C. P.; Adams, A. K.; Simpson, T.; Jr, R. E. P.; Moore, M. A . Treatment of bioprosthetic heart valve tissue with long chain alcohol solution to lower calcification potential . J. Biomed. Mater. Res. , 2004 . 69 140 -144 . DOI:10.1002/jbm.a.20129 .
Park, C. S.; Kim, Y. J.; Lee, J. R.; Lim, H. G.; Chang, J. E.; Jeong, S.; Kwon, N. . Anticalcification effect of a combination of decellularization, organic solvents and amino acid detoxification on glutaraldehyde-fixed xenopericardial heart valves in a large-animal long-term circulatory model . Interact. Cardiovasc. Thorac. Surg. , 2017 . 25 391 -399 . DOI:10.1093/icvts/ivx131 .
Rybinski, W. V.; Hill, K . Alkyl polyglycosides properties and applications of a new class of surfactants . Angew. Chem. Int. Ed. , 1998 . 37 1328 -1345 . DOI:10.1002/(SICI)1521-3773(19980605)37:10<1328::AID-ANIE1328>3.3.CO;2-0 .
Stoops, J. K.; Arora, R.; Armitage, L.; Wanger, A.; Song, L.; Blackburn, M. R.; Krueger, G. R.; Risin, S. A . Certain surfactants show promise in the therapy of pulmonary tuberculosis . In vivo , 2010 . 24 687 -694 . DOI:10.1089/hum.2009.171 .
Loke, W. K.; Khor, E . Validation of the shrinkage temperature of animal tissue for bioprosthetic heart valve application by differential scanning calorimetry . Biomoterials , 1995 . 251 -258 . DOI:10.1016/0142-9612(95)92125-P .
Kramer, R. Z.; Bella, J.; Brodsky, B.; Mayville, P.; Berman, H. M . Sequence dependent conformational variations of collagen triple-helical structure . Nat. Struct. Biol. , 1999 . 6 454 -457 . DOI:10.1038/8259 .
Vyazovkin, S.; Vincent, L.; Sbirrazzuoli, N . Thermal denaturation of collagen analyzed by isoconversional method . Macromol. Biosci. , 2007 . 7 1181 -1186 . DOI:10.1002/mabi.200700162 .
Shah, M. U. H.; Moniruzzaman, M.; Sivapragasam, M.; Talukder, M. M. R.; Yusup, S. B.; Goto, M . A binary mixture of a biosurfactant and an ionic liquid surfactant as a green dispersant for oil spill remediation . J. Mol. Liq. , 2019 . 280 111 -119 . DOI:10.1016/j.molliq.2019.02.049 .
Jin, D.; Jiang, X.; Jing, X.; Ou, Z . Effects of concentration, head group, and structure of surfactants on the degradation of phenanthrene . J. Hazard. Mater. , 2007 . 144 215 -221 . DOI:10.1016/j.jhazmat.2006.10.012 .
Singh, R. P.; Gupta, N.; Singh, S.; Singh, A.; Suman, R.; Annie, K. . Toxicity of ionic and nonionic surfactants to six macrobes found in Agra, India . Bull. Environ. Contam. Toxicol. , 2002 . 69 265 -270 . DOI:10.1007/s00128-002-0056-z .
Birk, D. E.; Bruckner, P. Collagen suprastructures. In Collagen: primer in structure, processing and assembly, Brinckmann, J.; Notbohm, H.; Müller, P. K., Eds. Springer, Berlin, 2005; pp. 185−205.
Mienaltowski, M. J.; Gonzales, N. L.; Beall, J. M.; Pechanec, M. Y . Basic structure, physiology, and biochemistry of connective tissues and extracellular matrix collagens . Adv. Exp. Med. Biol. , 2021 . 1348 5 -43 . DOI:10.1007/978-3-030-80614-9_2 .
Xie, Y.; Hu, C.; Feng, Y.; Li, D.; Ai, T.; Huang, Y.; Chen, X.; Huang, L.; Tan, J . Osteoimmunomodulatory effects of biomaterial modification strategies on macrophage polarization and bone regeneration . Regen. Biomater. , 2020 . 7 233 -245 . DOI:10.1093/rb/rbaa006 .
Luo, H.; Liu, W.; Zhou, Y.; Jiang, X.; Liu, Y.; Yang, Q.; Shao, L . Concentrated growth factor regulates the macrophage-mediated immune response . Regen. Biomater. , 2021 . rbab049 DOI:10.1093/rb/rbab049 .
Green, J. J.; Elisseeff, J. H . Mimicking biological functionality with polymers for biomedical applications . Nature , 2016 . 540 386 -394 . DOI:10.1038/nature21005 .
Xu, D.; Huang, J.; Zhao, D.; Ding, B.; Zhang, L.; Cai, J . High-flexibility, high-toughness double-cross-linked chitin hydrogels by sequential chemical and physical cross-linkings . Adv. Mater. , 2016 . 28 5844 -5849 . DOI:10.1002/adma.201600448 .
Du, J.; Wang, J. H.; Yu, H. Y.; Zhang, Y. Y.; Pu, L. H.; Wang, J. C.; Lu, S. Y.; Chen, S. H.; Zhu, T. H . Electrospun poly(p-dioxanone)/poly(ester-urethane)ureas composite nanofibers for potential heart valve tissue reconstruction . Chinese J. Polym. Sci. , 2019 . 37 560 -569 . DOI:10.1007/s10118-019-2231-2 .
Zhang, C.; Lu, H . Helical nonfouling polypeptides for biomedical applications . Chinese J. Polym. Sci. , 2022 . 40 433 -446 . DOI:10.1007/s10118-022-2688-2 .
Xu, X.; Gao, J.; Liu, S.; Chen, L.; Chen, M.; Yu, X.; Ma, N.; Zhang, J.; Chen, X.; Zhong, L.; Yu, L.; Xu, L.; Guo, Q.; Ding, J . Magnetic resonance imaging for non-invasive clinical evaluation of normal and regenerated cartilage . Regen. Biomater. , 2021 . 8 rbab038 DOI:10.1093/rb/rbab038 .
Murphy, S. V.; Atala, A . 3D bioprinting of tissues and organs . Nat. Biotechnol. , 2014 . 32 773 -785 . DOI:10.1038/nbt.2958 .
Bai, L.; Zhao, J.; Wang, M.; Feng, Y.; Ding, J . Matrix-metalloproteinase-responsive gene delivery surface for enhanced in situ endothelialization . ACS Appl. Mater. Interfaces , 2020 . 12 40121 -40132 . DOI:10.1021/acsami.0c11971 .
Yuan, Z.; Lin, C.; He, Y.; Tao, B.; Chen, M.; Zhang, J.; Liu, P.; Cai, K . Near-infrared light-triggered nitric-oxide-enhanced photodynamic therapy and low-temperature photothermal therapy for biofilm elimination . ACS Nano , 2020 . 14 3546 -3562 . DOI:10.1021/acsnano.9b09871 .
Cui, S.; Chen, L.; Yu, L.; Ding, J . Synergism among polydispersed amphiphilic block copolymers leading to spontaneous physical hydrogelation upon heating . Macromolecules , 2020 . 53 7726 -7739 . DOI:10.1021/acs.macromol.0c01430 .
Cui, S.; Yu, L.; Ding, J . Strategy of “block blends” to generate polymeric thermogels versus that of one-component block copolymer . Macromolecules , 2020 . 53 11051 -11064 . DOI:10.1021/acs.macromol.0c02488 .
Cui, S.; Wei, Y.; Bian, Q.; Zhu, Y.; Chen, X.; Zhuang, Y.; Cai, M.; Tang, J.; Yu, L.; Ding, J . Injectable thermogel generated by the "block blend" strategy as a biomaterial for endoscopic submucosal dissection . ACS Appl. Mater. Interfaces , 2021 . 13 19778 -19792 . DOI:10.1021/acsami.1c03849 .
Yu, X.; Li, G.; Zheng, Y.; Gao, J.; Fu, Y.; Wang, Q.; Huang, L.; Pan, X.; Ding, J . 'Invisible' orthodontics by polymeric 'clear' aligners molded on 3D-printed personalized dental models . Regen. Biomater. , 2022 . 9 rbac007 DOI:10.1093/rb/rbac007 .
Cai, C.; Tang, J.; Zhang, Y.; Rao, W.; Cao, D.; Guo, W.; Yu, L.; Ding, J . Intelligent paper-free sprayable skin mask based on an in situ formed janus hydrogel of an environmentally friendly polymer . Adv. Healthc. Mater. , 2022 . e2102654 DOI:10.1002/adhm.202102654 .
Rajamannan, N. M.; Evans, F. J.; Aikawa, E.; Grande-Allen, K. J.; Demer, L. L.; Heistad, D. D.; Simmons, C. A.; Masters, K. S.; Mathieu, P.; O'Brien, K. D.; Schoen, F. J.; Towler, D. A.; Yoganathan, A. P.; Otto, C. M. . Calcific aortic valve disease: not simply a degenerative process: a review and agenda for research from the National Heart and Lung and Blood Institute Aortic Stenosis Working Group. Executive summary: calcific aortic valve disease-2011 update . Circulation , 2011 . 124 1783 -1791 . DOI:10.1161/CIRCULATIONAHA.110.006767 .
Yao, X.; Ding, J . Effects of microstripe geometry on guided cell migration . ACS Appl. Mater. Interfaces , 2020 . 12 27971 -27983 . DOI:10.1021/acsami.0c05024 .
Liu, Q.; Zheng, S.; Ye, K.; He, J.; Shen, Y.; Cui, S.; Huang, J.; Gu, Y.; Ding, J . Cell migration regulated by RGD nanospacing and enhanced under moderate cell adhesion on biomaterials . Biomaterials , 2020 . 263 120327 DOI:10.1016/j.biomaterials.2020.120327 .
Yao, X.; Wang, X.; Ding, J . Exploration of possible cell chirality using material techniques of surface patterning . Acta Biomater. , 2021 . 126 92 -108 . DOI:10.1016/j.actbio.2021.02.032 .
Mao, T.; He, Y.; Gu, Y.; Yang, Y.; Yu, Y.; Wang, X.; Ding, J . Critical frequency and critical stretching rate for reorientation of cells on a cyclically stretched polymer in a microfluidic chip . ACS Appl. Mater. Interfaces , 2021 . 13 13934 -13948 . DOI:10.1021/acsami.0c21186 .
Zheng, S.; Liu, Q.; He, J.; Wang, X.; Ye, K.; Wang, X.; Yan, C.; Liu, P.; Ding, J . Critical adhesion areas of cells on micro-nanopatterns . Nano Res. , 2021 . 15 1623 -1635 . DOI:10.1007/s12274-021-3711-6 .
He, J.; Liu, Q.; Zheng, S.; Shen, R.; Wang, X.; Gao, J.; Wang, Q.; Huang, J.; Ding, J . Enlargement, reduction, and even reversal of relative migration speeds of endothelial and smooth muscle cells on biomaterials simply by adjusting RGD nanospacing . ACS Appl. Mater. Interfaces , 2021 . 13 42344 -42356 . DOI:10.1021/acsami.1c08559 .
Dalby, M. J.; Gadegaard, N.; Oreffo, R. O . Harnessing nanotopography and integrin-matrix interactions to influence stem cell fate . Nat. Mater. , 2014 . 13 558 -569 . DOI:10.1038/NMAT3980 .
Xu, W. K.; Tang, J. Y.; Yuan, Z.; Cai, C. Y.; Chen, X. B.; Cui, S. Q.; Liu, P.; Yu, L.; Cai, K. Y.; Ding, J. D . Accelerated cutaneous wound healing using an injectable teicoplanin-loaded PLGA-PEG-PLGA thermogel dressing . Chinese J. Polym. Sci. , 2019 . 37 548 -559 . DOI:10.1007/s10118-019-2212-5 .
Li, X.; Zhang, W.; Lin, W.; Qiu, H.; Qi, Y.; Ma, X.; Qi, H.; He, Y.; Zhang, H.; Qian, J.; Zhang, G.; Gao, R.; Zhang, D.; Ding, J . Long-term efficacy of biodegradable metal-polymer composite stents after the first and the second implantations into porcine coronary arteries . ACS Appl. Mater. Interfaces , 2020 . 12 15703 -15715 . DOI:10.1021/acsami.0c00971 .
Ding, X.; Gao, J.; Yu, X.; Shi, J.; Chen, J.; Yu, L.; Chen, S.; Ding, J . 3D-printed porous scaffolds of hydrogels modified with tgf-beta1 binding peptides to promote in vivo cartilage regeneration and animal gait restoration . ACS Appl. Mater. Interfaces , 2022 . 14 15982 -15995 . DOI:10.1021/acsami.2c00761 .
Tu, C. X.; Gao, C. Y . Recent advances on surface-modified biomaterials promoting selective adhesion and directional migration of cells . Chinese J. Polym. Sci. , 2021 . 39 815 -823 . DOI:10.1007/s10118-021-2564-5 .
Whelan, A.; Williams, E.; Fitzpatrick, E.; Murphy, B. P.; Gunning, P. S.; O'Reilly, D.; Lally, C. . Collagen fibre-mediated mechanical damage increases calcification of bovine pericardium for use in bioprosthetic heart valves . Acta Biomater. , 2021 . 128 384 -392 . DOI:10.1016/j.actbio.2021.04.046 .
Xu, Y.; Nudelman, F.; Eren, E. D.; Wirix, M. J. M.; Cantaert, B.; Nijhuis, W. H.; Hermida-Merino, D.; Portale, G.; Bomans, P. H. H.; Ottmann, C.; Friedrich, H.; Bras, W.; Akiva, A.; Orgel, J.; Meldrum, F. C.; Sommerdijk, N . Intermolecular channels direct crystal orientation in mineralized collagen . Nat. Commun. , 2020 . 11 1 -12 . DOI:10.1038/s41467-020-18846-2 .
Jones, M.; Eidbo, E. E.; Hilbert, S. L.; Ferrans, V. J.; Clark, R. E . Anticalcification treatments of bioprosthetic heart valves: in vivo studies in sheep . J. Card. Surg. , 1989 . 4 69 -73 . DOI:10.1111/j.1540-8191.1989.tb00258.x .
Agathos, E. A.; Tomos, P. I.; Kostomitsopoulos, N.; Koutsoukos, P. G . A novel anticalcification treatment strategy for bioprosthetic valves and review of the literature . J. Card. Surg. , 2019 . 34 895 -900 . DOI:10.1111/jocs.14151 .
Gao, J.; Yu, X.; Wang, X.; He, Y.; Ding, J . Biomaterial-related cell microenvironment in tissue engineering and regenerative medicine . Engineering , 2022 . 10 2001404 .
Li, B.; Xie, Z.; Wang, Q.; Chen, X.; Liu, Q.; Wang, W.; Shen, Y.; Liu, J.; Li, A.; Li, Y.; Zhang, G.; Liu, J.; Zhang, D.; Liu, C.; Wang, S.; Xie, Y.; Zhang, Z.; Ding, J . Biodegradable polymeric occluder for closure of atrial septal defect with interventional treatment of cardiovascular disease . Biomaterials , 2021 . 274 120851 DOI:10.1016/j.biomaterials.2021.120851 .
Pan, Z.; Duan, P.; Liu, X.; Wang, H.; Cao, L.; He, Y.; Dong, J.; Ding, J . Effect of porosities of bilayered porous scaffolds on spontaneous osteochondral repair in cartilage tissue engineering . Regen. Biomater. , 2015 . 2 9 -19 . DOI:10.1093/rb/rbv001 .
Gao, J.; Ding, X.; Yu, X.; Chen, X.; Zhang, X.; Cui, S.; Shi, J.; Chen, J.; Yu, L.; Chen, S.; Ding, J . Cell-free bilayered porous scaffolds for osteochondral regeneration fabricated by continuous 3D-printing using nascent physical hydrogel as ink . Adv. Healthc. Mater. , 2021 . 10 e2001404 DOI:10.1002/adhm.202001404 .
Shen, Y.; Zhang, W.; Xie, Y.; Li, A.; Wang, X.; Chen, X.; Liu, Q.; Wang, Q.; Zhang, G.; Liu, Q.; Liu, J.; Zhang, D.; Zhang, Z.; Ding, J . Surface modification to enhance cell migration on biomaterials and its combination with 3D structural design of occluders to improve interventional treatment of heart diseases . Biomaterials , 2021 . 279 121208 DOI:10.1016/j.biomaterials.2021.121208 .
Wang, X.; Lei, X.; Yu, Y.; Miao, S.; Tang, J.; Fu, Y.; Ye, K.; Shen, Y.; Shi, J.; Wu, H.; Zhu, Y.; Yu, L.; Pei, G.; Bi, L.; Ding, J . Biological sealing and integration of a fibrinogen-modified titanium alloy with soft and hard tissues in a rat model . Biomater. Sci. , 2021 . 9 5192 -5208 . DOI:10.1039/d1bm00762a .
Aulmann, W.; Sterzel, W. Toxicology of alkyl polyglycosides. in Alkyl Polyglycosides: Technology, Properties and Applications, VCH, Weinheim, New York, 1997, 151−167.
Wu, B.; Jin, L.; Ding, K.; Zhou, Y.; Yang, L.; Lei, Y.; Guo, Y.; Wang, Y . Extracellular matrix coating improves the biocompatibility of polymeric heart valves . J. Mater. Chem. B , 2020 . 8 10616 -10629 . DOI:10.1039/d0tb01884h .
32
Views
205
Downloads
6
CSCD
Related Articles
Related Author
Related Institution
