Fig 1 (a) 13C-NMR spectra and (b) FTIR spectra of initial CNF and CNF modified with a copolymer of SA and AMPS.

Ion-conducting Membranes Based on Bacterial Cellulose Nanofibers Modified by Poly(sodium acrylate-
co -2-acrylamido-2-methylpropanesulfonic acid)Ion-conducting Membranes Based on Bacterial Cellulose Nanofibers Modified by Poly(sodium acrylate-
co -2-acrylamido-2-methylpropanesulfonic acid)- 高分子科学(英文版) 2024年42卷第3期 页码:333-343
DOI:10.1007/s10118-023-3054-8
扫 描 看 全 文
引用本文
阅读全文PDF
HTML全文
图(8) 表(0)
参考文献
出版信息
数据统计
Abstract
Green method for preparation of ion-conducting membranes (ICM) based on bacterial cellulose nanofibers (CNF) modified by a copolymer of sodium acrylate and 2-acrylamido-2-methylpropanesulfonic acid was elaborated. FTIR and NMR data confirmed grafting of polyacrylate onto cellulose surface. Formation of porous structure of the ICM was controlled by SEM and AFM. The maximal ionic conductivity of the membranes reaches 1.5 and 3.1 mS·cm−1 (60 °C and 98% relative humidity) when they are saturated with water or H2SO4 (1 mol·L−1) electrolyte, respectively. Prepared ICM was tested as a separator in a symmetrical supercapacitor with electrodes based on polyaniline hydrogel. The assembled cell demonstrate ability to operate at high current density up to 100 A·g−1 maintaining specific capacitance 165 F·g−1. Maximal specific capacitance of 289 F·g−1 was achieved at current density 1 A·g−1. Retaining of 90% of initial capacitance after 10000 of charge-discharge cycles proves high electrochemical stability of prepared ICM.
摘要
Green ion-conducting membranes based on bacterial cellulose nanofibers with grafted acrylic copolymer was elaborated. High ion mobility and low activation energy of conductivity of prepared membrane allow supercapacitor cell to operate at an ultra-high current density.
INTRODUCTION
Due to increasing of energy consumption and growing number of portable devices, the development of eco-friendly and sustainable materials for energy storage[
Cellulose is the most abundant biopolymer,[
The way that is often used for preparation of BC based ICM is mixing it with other polymer that contains ionogenic groups. For example, membranes prepared by mixing of BC and Nafion with subsequent casting demonstrated heat resistance, high mechanical strength and exhibited the maximum ionic conductivity of 71 mS·cm−1 at 30 °C and 100% relative humidity (RH).[
Additional benefit of cellulose is the availability of reactive hydroxyl groups on its surface. This allows chemical modification in order to introduce ionogenic groups providing the appearance of mobile ions inside the membrane and thus regulating of ionic conductivity.[
The widely used industrial sulphonated polymer is prepared by polymerization of 2-acrylamido-2-methyl-1-propanesulfonic acid (AMPS) and attracts much attention due to its very high degree of dissociation, low price, non-toxicity, hydrolytic stability and swelling behavior.[
EXPERIMENTAL
Materials
The lyophilized culture of A. xylinum was purchased from the All-Russian collection of industrial microorganisms (National Bioresource Center, GosNIIgenetics, Moscow, Russia) and cultured. Peptone and D-mannitol (CAS 69-65-8) were obtained from LenReaktiv (Saint Petersburg, Russia). Choline chloride (ChCl) (Glentham Life Sciences Ltd., Corsham, UK, CAS 67-48-1, purity>99%) was dried under vacuum at 60 °C for at least 24 h before use. Urea (LenReactiv, Saint Petersburg, Russia, CAS 57-13-6, purity 98%). AMPS (CAS 15214-89-8) and acrylic acid (AA) (CAS 607-061-00-8, purity >99%) were purchased from Merck, German and from Sigma-Aldrich, Czech Republic, respectively. Cerium (IV) ammonium nitrate ((NH4)2Ce(NO3)6) (Merck, German, CAS16774-21-3), nitric acid (HNO3) (Vekton, Saint Petersburg, Russia, CAS 7697-37-2) and sodium chloride (Vekton, Saint Petersburg, Russia) were used.
Preparation of Bacterial Cellulose Nanofibers
CNF were obtained according to a previously developed method.[
Chemical Modification of Bacterial Cellulose Nanofibers
Dispersion of CNF was used as a raw material for modification. Grafting of poly(sodium acrylate-co-2-acrylamido-2-methylpropanesulfonic acid) (SA-AMPS) was performed on the surface of CNF using cerium (IV) ammonium nitrate ((NH4)2Ce(NO3)6) as initiator under optimal conditions found during preliminary studies (see Table S1 in the electronic supplementary information, ESI). In the case of optimal conditions АА (1.296 g) was mixed with equimolar amount of NaOH (0.72 g) and 2 mL of H2O in a beaker at room temperature. Then AMPS was dissolved in 8 mL of H2O and added to the solution of sodium acrylate in amount of 8.694 g. A solution of 0.2467 g of (NH4)2Ce(NO3)6 and 0.355 g of HNO3 was prepared in 2 mL of H2O and added to 7.5 g the CNF dispersion (2 wt%). Then the solution of monomers was added. The polymerization was carried out at 60 °C for 4 h. The resulting mixture was kept for 12 h at room temperature. The product was purified from not-grafted acrylic polymer by repeating of the dispersion-centrifugation procedures at least 10 times at a speed of 6000 r/min.
Preparation of Membranes from Modified Bacterial Cellulose
The obtained dispersions were diluted with water in order to attain a 0.3 wt% concentration of CNF(M). Sodium chloride (NaCl) as an agent for regulation of porosity[
The densities of prepared membranes were calculated from their mass measured gravimetrically using an AP225WD (Shimadzu, Kyoto, Japan) microbalance using pieces 20 nm × 20 mm and measured thickness that was 16−24 µm. The porosity (P) of membranes was estimated from density measurements as P = 100 × (ρnon-p – ρp)/ρnon-p, where ρnon-p is the density of non-porous film (CNF(M)-0 in our case) and ρp is the density of porous one.
Spectroscopy Study
Chemical structures of CNF and CNF(M) were characterized using IRAffinity-1S spectrometer (Shimadzu, Kyoto, Japan), in attenuated total reflectance (ATR) mode, with a scan range from 4000 cm−1 to 600 cm−1. A total of 100 scans were made with a resolution of 2 cm−1. Additional information was obtained using 13C-NMR study using AVANCE II-500 WB NMR Fourier Spectrometer (Bruker, Germany).
Microscopic Investigation
Atomic force microscopy (AFM) studies were performed using SPM-9700HT scanning probe microscope (Shimadzu, Kyoto, Japan) in the tapping mode. NSG30-SS silicon cantilevers with a probe radius of 2 nm were used. Scanning electron microscopy (SEM) images of the membranes’ surfaces were obtained with SUPRA 55 VP microscope (Carl Zeiss, Oberkochen, Germany) at voltage of 5 kV. Content of ―SO3H groups in the CNF(M) was determined with energy dispersive X-ray (EDX) spectroscopy using Aztec Energy X-act microanalysis system (Oxford Instruments, UK) of Tescan Vega III SEM microscope (Tescan, Brno-Kohoutovice, Czech Republic). Transmission electron microscopy (TEM) studies were performed using JEM-2200 FS (JEOL, Japan).
Wide-angle X-ray Diffraction Study
The crystalline structure of the CNF and CNF(M) was studied via wide-angle X-ray diffraction (WAXD) with Rigaku SmartLab 3 diffractometer (Rigaku Corporation, Tokyo, Japan) equipped with CuKα radiation source (λ=1.54 Å) within the 2θ range of 5°−40° with the scan step of 0.05°.
Measurement of Water Sorption Isotherms of Membranes
Sorption isotherms of the membranes were determined gravimetrically at room temperature. Six saturated salt solutions were selected, CaCl2, NaNO3, MnCl2, NaCl, KCl and K2SO4 (RH=38%, 56.3%, 62.6%, 84.9%, 88.3% and 96.2%, respectively). RH value was controlled with an IVTM-7 K thermohygrometer (Moscow, Russia). The membranes were dried under vacuum at 60 °C for 7 days before experiments to remove residual moisture. Samples were placed in preliminarily weighed plastic sorption jars, and then kept inside desiccators with fixed RH. Masses of membranes were measured every 24 h until the constant values were reached. The amount of absorbed water was calculated and sorption isotherms were plotted. The Laatikainen-Lindstrom model[
a=amαh(1−βh)[1+(α−β)h] 1
where am and h are the concentration of primary sorption centers and the ratio of the partial pressure to the saturated vapor pressure. Based on α and β parameters the sorption on primary sorption centers (polar groups of polymer) and on secondary sorption centers (on previously sorbed water molecules, so called sorption in clusters) was estimated.
Mechanical Measurements
The stress-strain curves for the membranes were measured using the AG-100kNX Plus (Shimadzu, Japan) machine for mechanical measurements operating in the uniaxial extension mode. The ambient conditions during the experiment were 25 °C and 75% of relative humidity. Strip-like samples (2 mm × 15 mm) were clamped in the analyzer, and the load (N) and elongation (mm) were recorded with an extension velocity of 0.5 mm per minute. The values of Young’s modulus, elongation at break and ultimate strain were obtained.
Electrochemical Measurements
All electrochemical measurements were conducted under the constant RH 98% using a climate chamber ERSTEVAK KTXB-64-D (Moscow, Russia) coupled with a P-40X potentiostat/galvanostat equipped with FRA24 impedance module (Elins, Moscow, Russia). The ionic conductivity of the membranes was studied over a range of temperatures 20−60 °C. The impedance spectra were acquired from 10 Hz to 100 kHz at voltage amplitude of 10 mV. Before measurement the membrane was soaked in distilled water or H2SO4 solution with concentration 1 mol·L−1. The membrane resistance (R) was determined from the high frequency intercept of the Nyquist plot with the axis of the real part of complex impedance. The through-plane conductivity σ (mS·cm−1) was calculated using the following equation:
σ=LRS 2
where R is the electrical resistance, L is the membrane thickness and S is the electrode area.
In order to demonstrate practical applicability of prepared membranes, the model symmetric supercapacitor (SC) cell was built. For this purpose, the polyacrylamide-polyaniline (PAAm-PANI) based electrodes were prepared according to the method described earlier.[
The electrochemical characteristics of prepared SC were investigated with cyclic voltammetry (CVA), galvanostatic charge-discharge (GCD), electrochemical impedance spectroscopy (EIS) and cycle stability measurements. The specific capacitance of SC was calculated from the discharge branch of GCD curves as CS = 2 × It/(dU·m), where I and t are the discharge current and duration, respectively; dU is the voltage range and m is the mass of active material on one electrode.
RESULTS AND DISCUSSION
Modification of CNF and Their Spectroscopic Characterization
It is known that (NH4)2Ce(NO3)6 is able to interact with organic compounds containing ―OH groups with formation of radicals on organic molecule that are able to initiate polymerization of acrylate monomers. The possible mechanisms of formation of such radical in the case of reaction with cellulose can be found elsewhere.[
However, it is seen from 13C-NMR results that the modification of cellulose did not happen: peaks corresponding to acrylic monomer cannot be found in NMR spectrum (see Fig. S1 in ESI). Only the peaks corresponding to cellulose at 102−108 (C1), 80−92 (C4), 70−80 (C2, C3 and C5), 57−67 ppm (C6) are visible (see Fig. S1 in ESI, CNF(M1) sample). Carbon atoms in brackets denotes numbers of atoms in glycoside ring that correspond to the mentioned peaks according to literature.[
Additional study of chemical structure of modified CNF was performed with FTIR (
Investigation of Morphology of CNF(M) and ICM
The detailed images of the internal structures of CNF and CNF(M) were investigated by TEM (see
Fig 2 TEM images for (a) initial and (b) modified CNF; (c) WAXD patterns for CNF and CNF(M).
The structure of ICM prepared by casting of CNF(M) dispersions was studied with SEM and AFM (
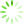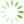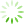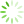
Fig 3 SEM (a, b) and AFM images (c, d) for non-porous (a, c) and porous (b, d) ICM based on CNF(M).
Measurement of Water Sorption Isotherms of ICM
Data on the water sorption activity of prepared ICM gives additional information about the physico-chemical properties of active sorption centers in polymer and behavior of materials under different humidity. The water sorption values of ICM at different RH were measured using the static gravimetric method (result is shown as points in
Fig 4 Experimental values of water sorption for prepared ICM (points) and the result of their approximation by the LL isotherm (lines).
| Sample | am | α | β |
|---|---|---|---|
| pristine CNF | 0.1245 | 2.1125 | 0.8128 |
| CNF(M)-0 | 0.3164 | 1.8167 | 0.8629 |
| CNF(M)-20 | 0.4898 | 73.9812 | 0.9330 |
Higher values of parameter am for CNF(M)-20 membrane in comparison with CNF(M)-0 and membrane based on pristine CNF (
Fig 5 Dependences of water sorption on the primary sorption centers of the polymer (a) and in clusters (b) on the activity of water vapor.
Mechanical Measurements
The stress-strain curves measured in uniaxial tension mode are shown in
Fig 6 Stress-strain curves for non-porous CNF(M)-0 and porous membrane CNF(M)-20.
Electrochemical Studies
Ionic conductivity
For measurement of electrochemical properties, prepared ICM were saturated with distilled water or with aqueous solution of H2SO4 (1 mol·L−1). Throughout the following discussion, the corresponding membranes will be denoted as CNF(M)-X/H2O and CNF(M)-X/H2SO4. The curves of the electrochemical impedance for obtained ICM are given in
Fig 7 Electrochemical impedance spectra for CNF(M)-0/H2O, CNF(M)-20/H2O and CNF(M)-20/H2SO4 (a) and dependencies of ionic conductivity of ICM on reciprocal temperature with determined activation energies of ionic conductivity (b).
Values of ionic conductivity of prepared ICM in a temperature range of 20−60 °C are given in Fig. S4 (in ESI). The maximal value obtained for CNF(M)-20/H2O is 1.52 mS·cm−1 at 60 °C that is comparable to the values for ICM based on BC and fucoidan with conductivity 1.6 mS·cm−1[
In the case of CNF(M)-20/H2SO4 increase in ionic conductivity up to 1.79 mS·cm−1 at room temperature (about 2 times in comparison with water saturated ICM) and insignificant reduction of the activation energy down to 11.0 kJ·mol−1 were observed (
Testing of model supercapacitor cell
For testing of model supercapacitor cell, the porous CNF(M)-20 membrane was soaked in the 1 mol·L−1 solution of H2SO4 or water and then sandwiched between two PAAm-PANI electrodes. The cell was initially tested by the CVA in the voltage range of 0−600 mV and at the scan rate from 10 mV·s−1 to 100 mV·s−1. Measured currents were normalized by the mass of active material in the electrodes (PANI). It can be seen in
Fig 8 CV curves of (a) CNF(M)-20/H2SO4-based and (b) CNF(M)-20/H2O-based SC cells, GCD curves of (c) CNF(M)-20/H2SO4-based and (d) CNF(M)-20/H2O-based SC cells, (e) dependences of specific capacitance on current density and (f) capacitance retention after GCD cycling at 10 A·g−1.
It can be hypothesized that the increase in capacitance can be connected with hydrogel nature electroactive electrodes used for preparation of model cell in this study. In hydrogel state (being swollen with electrolyte) the electroconducting polymer chains are more mobile than in the case of solid porous electrode and thus conformational changes caused by chain movement during repetitive oxidation-reduction cycles can result in increasing availability of PANI chains for electrochemical reactions. The same effect was observed earlier for similar electrodes.[
CONCLUSIONS
The successful grafting of ionogenic acrylic copolymer on to the surface of CNF using cerium(IV) ammonium nitrate as initiator was demonstrated for the first time. The chemical structure of prepared nanomaterial was confirmed with NMR, FTIR and EDX studies. TEM results show the formation of an amorphous shell of polyacrylate onto a highly crystalline cellulose backbone. The possibility of preparation of self-standing porous ion-conducting membranes (ICM) via casting from dispersion of modified CNF was shown. Elaborated ICM had a high ionic conductivity up to 1.79 mS·cm−1 at room temperature and 3.1 mS·cm−1 at 60 °C due to porous structure and effective modification by grafting of ionogenic polyacrylate. In spite of moderate mechanical characteristics of prepared membranes, the self-standing properties were enough to use them as a separator in a symmetrical supercapacitor with polyacrylamide-polyaniline based electrodes. Due to high ion mobility and low activation energy of ionic conductivity, the supercapacitor cell based on prepared membranes demonstrated the possibility to operate at a high current density up to 100 A·g−1 maintaining 165 F·g−1 of specific capacitance. The maximal specific capacitance was achieved for CNF(M)-20/H2SO4-based cell: 289 F·g−1 at 1 A·g−1. In addition, cells show very high electrochemical stability during 10000 of charge-discharge cycles with water or acidic electrolyte retaining 88% and 90% of initial capacitance, respectively.
References
deOliveira,C.C.N.;Angelkorte,G.;Rochedo,P.R.R.;Szklo,A.Theroleofbiomaterialsfortheenergytransitionfromthelensofanationalintegratedassessmentmodel.Clim. Change2021, 167,57.. [百度学术]
Jian,M.;Zhang,Y.;Liu,Z.Naturalbiopolymersforflexiblesensingandenergydevices.Chinese J. Polym. Sci.2020, 38,459−490.. [百度学术]
Wang,Y.;RuizDiaz,D.F.;Chen,K.S.;Wang,Z.;Adroher,X.C.Materials,technologicalstatus,andfundamentalsofPEMfuelcells—areview.Mater. Today2020, 32,178−203.. [百度学术]
Lizundia,E.;Kundu,D.Advancesinnaturalbiopolymer-basedelectrolytesandseparatorsforbatteryapplications.Adv. Funct. Mater.2021, 31,1−29.. [百度学术]
Wang,S.;Zhang,L.;Zeng,Q.;Liu,X.;Lai,W.Y.;Zhang,L.Cellulosemicrocrystalswithbrush-likearchitecturesasflexibleall-solid-statepolymerelectrolyteforlithium-ionbattery.ACS Sustainable Chem. Eng.2020, 8,3200−3207.. [百度学术]
Wang,S.;He,J.;Li,Q.;Wang,Y.;Liu,C.;Cheng,T.;Lai,W.Y.Highlyelasticenergystoragedevicebasedonintrinsicallysuper-stretchablepolymerlithium-ionconductorwithhighconductivity.Fundam. Res2022,DOI:10.1016/j.fmre.2022.06.003. [百度学术]
Wang,S.;Bai,M.;Liu,C.;Li,G.;Lu,X.;Cai,H.;Liu,C.;Lai,W.Y.Highlystretchablemultifunctionalpolymerionicconductorwithhighconductivitybasedonorganic-inorganicdualnetworks.Chem. Eng. J.2022, 440,135824.. [百度学术]
Okonkwo,P.C.;Collins,E.;Okonkwo,E.ApplicationofBiopolymerCompositesinSuperCapacitor,inBiopolymer Composites in Electronics,Elsevier, 2017 :bll487–503.. [百度学术]
Musa,M.T.;Shaari,N.;Kamarudin,S.K.;Wong,W.Y.Recentbiopolymersusedformembranefuelcells:characterizationanalysisperspectives.Int. J. Energy Res.2022, 46,16178−16207.. [百度学术]
Palanisamy,G.;Oh,T.H.;Thangarasu,S.Modifiedcelluloseproton-exchangemembranesfordirectmethanolfuelcells.Polymers 2023 ,15,639.. [百度学术]
Winter,M.;Brodd,R.J.Whatarebatteries,fuelcells,andsupercapacitors.Chem. Rev.2004, 104,4245−4269.. [百度学术]
Ng,W.W.;Thiam,H.S.;Pang,Y.L.;Chong,K.C.;Lai,S.O.Astate-of-artonthedevelopmentofnafion-basedmembraneforperformanceimprovementindirectmethanolfuelcells.Membranes 2022 ,12,S06.. [百度学术]
Xu,T.C.;Wang,C.S.;Hu,Z.Y.;Zheng,J.J.;Jiang,S.H.;He,S.J.;Hou,H.Q.Highstrengthandstableprotonexchangemembranebasedonperfluorosulfonicacid/polybenzimidazole.Chinese J. Polym. Sci.2022, 40,764−771.. [百度学术]
Walkowiak-Kulikowska,J.;Wolska,J.;Koroniak,H.Biopolymermembranesinfuelcellapplications,inBiopolymer Membranes and Films,Elsevier, 2020 :bll423–476.. [百度学术]
Bayer,T.;Cunning,B.V.;Šmíd,B.;Selyanchyn,R.;Fujikawa,S.;Sasaki,K.;Lyth,S.M.Spraydepositionofsulfonatedcellulosenanofibersaselectrolytemembranesinfuelcells.Cellulose2021, 28,1355−1367.. [百度学术]
Reddy,M.S.B.;Ponnamma,D.;Choudhary,R.;Sadasivuni,K.K.Acomparativereviewofnaturalandsyntheticbiopolymercompositescaffolds.Polymers 2021 ,13,1105.. [百度学术]
Selyanchyn,O.;Selyanchyn,R.;Lyth,S.M.Areviewofprotonconductivityincellulosicmaterials.Front. Energy Res.2020, 8,1−17.. [百度学术]
Smirnov,M.A.;Sokolova,M.P.;Bobrova,N.V.;Toikka,A.M.;Morganti,P.;Lahderanta,E.Synergisticeffectofchitinnanofibersandpolyacrylamideonelectrochemicalperformanceoftheirternarycompositewithpolypyrrole.J. Energy Chem.2018, 27,843−853.. [百度学术]
Vijayakumar,V.;Nam,S.Y.Areviewofrecentchitosananionexchangemembranesforpolymerelectrolytemembranefuelcells.Membranes2022, 12,1−12.. [百度学术]
Vorobiov,V.K.;Smirnov,M.A.;Bobrova,N.V.;Sokolova,M.P.Chitosan-supporteddeepeutecticsolventasbio-basedelectrolyteforflexiblesupercapacitor.Mater. Lett.2021, 283,128889.. [百度学术]
Wang,B.;Han,X.;Wang,Y.;Kang,L.;Yang,Y.;Cui,L.;Zhong,S.;Cui,X.Fabricationofalginate-basedmulti-crosslinkedbiomembranesfordirectmethanolfuelcellapplication.Carbohydr. Polym.2023, 300,120261.. [百度学术]
Tiwari,T.;Srivastava,N.Exploringthepossibilityofstarch-basedelectrolytemembraneinMFCapplication.Macromol. Symp.2019, 388,1−8.. [百度学术]
Mohanapriya,S.;Rambabu,G.;Bhat,S.D.;Raj,V.Pectinbasednanocompositemembranesasgreenelectrolytesfordirectmethanolfuelcells.Arab. J. Chem.2020, 13,2024−2040.. [百度学术]
Hernández-Flores,G.;Andrio,A.;Compañ,V.;Solorza-Feria,O.;Poggi-Varaldo,H.M.Synthesisandcharacterizationoforganicagar-basedmembranesformicrobialfuelcells.J. Power Sources 2019 ,435,. [百度学术]
KumarGupta,P.;SaiRaghunath,S.;VenkateshPrasanna,D.;Venkat,P.;Shree,V.;Chithananthan,C.;Choudhary,S.;Surender,K.;Geetha,K.Anupdateonoverviewofcellulose,itsstructureandapplications,inCellulose,IntechOpen, 2019 :bll1–21. [百度学术]
Heinze,T.;ElSeoud,O.A.;Koschella,A.Productionandcharacteristicsofcellulosefromdifferentsources, 2018 . [百度学术]
Gadim,T.D.O.;Loureiro,F.J.A.;Vilela,C.;Rosero-Navarro,N.;Silvestre,A.J.D.;Freire,C.S.R.;Figueiredo,F.M.L.Protonicconductivityandfuelcelltestsofnanocompositemembranesbasedonbacterialcellulose.Electrochim. Acta2017, 233,52−61.. [百度学术]
Batishcheva,E.V.;Sokolova,D.N.;Fedotova,V.S.;Sokolova,M.P.;Nikolaeva,A.L.;Vakulyuk,A.Y.;Shakhbazova,C.Y.;Ribeiro,M.C.C.;Karttunen,M.;Smirnov,M.A.Strengtheningcellulosenanopaperviadeepeutecticsolventandultrasound-inducedsurfacedisorderingofnanofibers.Polymers 2022 ,14,78.. [百度学术]
Wang,Y.R.;Yin,C.C.;Zhang,J.M.;Wu,J.;Yu,J.;Zhang,J.Functionalcellulosematerialsfabricatedbyusingionicliquidsasthesolvent.Chinese J. Polym. Sci.2023, 41,483−499.. [百度学术]
Lin,D.;Liu,Z.;Shen,R.;Chen,S.;Yang,X.Bacterialcelluloseinfoodindustry:currentresearchandfutureprospects.Int. J. Biol. Macromol.2020, 158,1007−1019.. [百度学术]
Smirnov,M.A.;Fedotova,V.S.;Sokolova,M.P.;Nikolaeva,A.L.;Elokhovsky,V.Y.;Karttunen,M.Polymerizablecholine-andimidazolium-basedionicliquidsreinforcedwithbacterialcellulosefor3D-printing.Polymers 2021 ,13,3044.. [百度学术]
Sathish,S.K.;Vitta,S.Bacterial Cellulose Based Nanocomposites for Electronic and Energy Applications.Elsevier. 2020 .. [百度学术]
Ramírez-Carmona,M.;Gálvez-Gómez,M.P.;González-Perez,L.;Pinedo-Rangel,V.;Pineda-Vasquez,T.;Hotza,D.Productionofbacterialcellulosehydrogelanditsevaluationasaprotonexchangemembrane.J. Polym. Environ. 2023 ,31,2462−2472.. [百度学术]
Smirnov,M.A.;Vorobiov,V.K.;Sokolova,M.P.;Bobrova,N.V.;Lahderanta,E.;Hiltunen,S.;Yakimansky,A.V.Electrochemicalpropertiesofsupercapacitorelectrodesbasedonpolypyrroleandenzymaticallypreparedcellulosenanofibers.Polym. Sci. - Ser. C.2018, 60,228−239.. [百度学术]
Shu,Y.;Bai,Q.;Fu,G.;Xiong,Q.;Li,C.;Ding,H.;Shen,Y.;Uyama,H.Hierarchicalporouscarbonsfrompolysaccharidescarboxymethylcellulose,bacterialcellulose,andcitricacidforsupercapacitor.Carbohydr. Polym.2020, 227,115346.. [百度学术]
Lahiri,D.;Nag,M.;Dutta,B.;Dey,A.;Sarkar,T.;Pati,S.;Edinur,H.A.;Kari,Z.A.;Noor,N.H.M.;Ray,R.R.Bacterialcellulose:production,characterizationandapplicationasantimicrobialagent.Int. J. Mol. Sci.2021, 22,1−18.. [百度学术]
Mishra,S.;Singh,P.K.;Pattnaik,R.;Kumar,S.;Ojha,S.K.;Srichandan,H.;Parhi,P.K.;Jyothi,R.K.;Sarangi,P.K.Biochemistry,synthesis,andapplicationsofbacterialcellulose:areview.Front. Bioeng. Biotechnol.2022, 10,1−12.. [百度学术]
Jiang,G.;Zhang,J.;Qiao,J.;Jiang,Y.;Zarrin,H.;Chen,Z.;Hong,F.Bacterialnanocellulose/Nafioncompositemembranesforlowtemperaturepolymerelectrolytefuelcells.J. Power Sources2015, 273,697−706.. [百度学术]
Gadim,T.D.O.;Vilela,C.;Loureiro,F.J.A.;Silvestre,A.J.D.;Freire,C.S.R.;Figueiredo,F.M.L.Nafion®andnanocellulose:apartnershipforgreenerpolymerelectrolytemembranes.Ind. Crops Prod.2016, 93,212−218.. [百度学术]
Samaniego,A.J.;Espiritu,R.Prospectsonutilizationofbiopolymermaterialsforionexchangemembranesinfuelcells.Green Chem. Lett. Rev.2022, 15,253−275.. [百度学术]
Dahlström,C.;LópezDurán,V.;Keene,S.T.;Salleo,A.;Norgren,M.;Wågberg,L.IonconductivitythroughTEMPO-mediatedoxidatedandperiodateoxidatedcellulosemembranes.Carbohydr. Polym.2020, 233,115829.. [百度学术]
Rana,H.H.;Park,J.H.;Gund,G.S.;Park,H.S.Highlyconducting,extremelydurable,phosphorylatedcellulose-basedionogelsforrenewableflexiblesupercapacitors.Energy Storage Mater.2020, 25,70−75.. [百度学术]
Yue,L.;Zheng,Y.;Xie,Y.;Liu,S.;Guo,S.;Yang,B.;Tang,T.Preparationofacarboxymethylatedbacterialcellulose/polyanilinecompositegelmembraneanditscharacterization.RSC Adv.2016, 6,68599−68605.. [百度学术]
Yue,L.;Xie,Y.;Zheng,Y.;He,W.;Guo,S.;Sun,Y.;Zhang,T.;Liu,S.Sulfonatedbacterialcellulose/polyanilinecompositemembraneforuseasgelpolymerelectrolyte.Compos. Sci. Technol.2017, 145,122−131.. [百度学术]
Sriruangrungkamol,A.;Chonkaew,W.Modificationofnanocellulosemembranebyimpregnationmethodwithsulfosuccinicacidfordirectmethanolfuelcellapplications.Polym. Bull.2021, 78,3705−3728.. [百度学术]
Hao,Y.;Qu,J.;Tan,L.;Liu,Z.;Wang,Y.;Lin,T.;Yang,H.;Peng,J.;Zhai,M.Synthesisandpropertyofsuperabsorbentpolymerbasedoncellulosegrafted2-acrylamido-2-methyl-1-propanesulfonicacid.Int. J. Biol. Macromol.2023, 233,123643.. [百度学术]
Anirudhan,T.S.;Rejeena,S.R.Poly(acrylicacid- co-acrylamide- co-2-acrylamido-2-methyl-1-propanesulfonicacid)-graftednanocellulose/poly(vinylalcohol)compositeforthe in vitrogastrointestinalreleaseofamoxicillin.J. Appl. Polym. Sci.2014, 131,8657−8668.. [百度学术]
Prosvirnina,A.P.;Bugrov,A.N.;Dobrodumov,A.V.;Vlasova,E.N.;Fedotova,V.S.;Nikolaeva,A.L.;Vorobiov,V.K.;Sokolova,M.P.;Smirnov,M.A.Bacterialcellulosenanofibersmodificationwith3-(trimethoxysilyl)propylmethacrylateasacrosslinkingandreinforcingagentfor3DprintableUV-curableinks.J. Mater. Sci.2022, 57,20543−20557.. [百度学术]
Fager,C.;Gebäck,T.;Hjärtstam,J.;Röding,M.;Olsson,A.;Lorén,N.;vonCorswant,C.;Särkkä,A.;Olsson,E.Correlating3DporousstructureinpolymerfilmswithmasstransportpropertiesusingFIB-SEMtomography.Chem. Eng. Sci. X.2021, 12,100109.. [百度学术]
Tessmar,J.;Holland,T.;Mikos,A.SaltLeachingforPolymerScaffolds,inScaffolding In Tissue Engineering,CRCPress, 2005 ,bll111–124. [百度学术]
Laatikainen,M.;Lindstrom,M.Generalsorptionisothermforswellingmaterials.Acta Polytech. Scand. Technol. Ser.1987, 178,105−116.. [百度学术]
Smirnov,M.A.;Sokolova,M.P.;Bobrova,N.V.;Kasatkin,I.A.;Lahderanta,E.;Elyashevich,G.K.Capacitancepropertiesandstructureofelectroconductinghydrogelsbasedoncopoly(aniline-P-phenylenediamine)andpolyacrylamide.J. Power Sources2016, 304,102−110.. [百度学术]
Gaylord,N.Proposednewmechanismforcatalyzedanduncatalyzedgraftpolymerizationontocellulose.J. Polym. Sci., Part C: Polym Symp.1972, 172,153−172.. [百度学术]
Kalia,S.;Sabaa,M.W.inPolysaccharide based graft copolymers,SpringerLink, 2013 .. [百度学术]
Jacek,P.;Kubiak,K.;Ryngajłło,M.;Rytczak,P.;Paluch,P.;Bielecki,S.ModificationofbacterialnanocellulosepropertiesthroughmutationofmotilityrelatedgenesinKomagataeibacterhanseniiATCC53582.N. Biotechnol.2019, 52,60−68.. [百度学术]
Gelenter,M.D.;Wang,T.;Liao,S.Y.;O’Neill,H.;Hong,M.2H-13Ccorrelationsolid-stateNMRforinvestigatingdynamicsandwateraccessibilitiesofproteinsandcarbohydrates.J. Biomol. NMR. 2017 ,68,257–270.. [百度学术]
Fedotova,V.S.;Sokolova,M.P.;Vorobiov,V.K.;Sivtsov,E.V.;Lukasheva,N.V.;Smirnov,M.A.Waterinfluenceonthephysico-chemicalpropertiesand3Dprintabilityofcholineacrylate—bacterialcelluloseinks.Polymers 2023 ,15,2156.. [百度学术]
Porwal,S.;Diwedi,A.;Kamal,M.13CNMRandRamanstudiesoffullerene-basedpoly(acrylamides).Int. J. Org. Chem. 2012 ,02,377–386.. [百度学术]
Devrim,Y.G.;Rzaev,Z.M.O.;Pişkin,E.Synthesisandcharacterizationofpoly[((maleicanhydride)- alt-styrene)- co-(2-acrylamido-2-methyl-1-propanesulfonicacid)].Macromol. Chem. Phys.2006, 207,111−121.. [百度学术]
Qiao,J.;Hamaya,T.;Okada,T.Newhighlyproton-conductingmembranepoly(vinylpyrrolidone)(PVP)modifiedpoly(vinylalcohol)/2-acrylamido-2-methyl-1-propanesulfonicacid(PVA-PAMPS)forlowtemperaturedirectmethanolfuelcells(DMFCs).Polymer2005, 46,10809−10816.. [百度学术]
Mondal,M.I.H.Mechanismofstructureformationofmicrobialcelluloseduringnascentstage.Cellulose2013, 20,1073−1088.. [百度学术]
Jiang,G.;Qiao,J.;Hong,F.Applicationofphosphoricacidandphyticacid-dopedbacterialcelluloseasnovelproton-conductingmembranestoPEMFC.Int. J. Hydrogen Energy.2012, 37,9182−9192.. [百度学术]
Ni,C.;Wang,H.;Zhao,Q.;Liu,B.;Sun,Z.;Zhang,M.;Hu,W.;Liang,L.Crosslinkingeffectinnanocrystallinecellulosereinforcedsulfonatedpoly(aryletherketone)protonexchangemembranes.Solid State Ionics2018, 323,5−15.. [百度学术]
Selyanchyn,O.;Bayer,T.;Klotz,D.;Selyanchyn,R.;Sasaki,K.;Lyth,S.M.Cellulosenanocrystalscrosslinkedwithsulfosuccinicacidassustainableprotonexchangemembranesforelectrochemicalenergyapplications.Membranes 2022 ,12,658.. [百度学术]
Gadim,T.D.O.;Figueiredo,A.G.P.R.;Rosero-Navarro,N.C.;Vilela,C.;Gamelas,J.A.F.;Barros-Timmons,A.;Neto,C.P.;Silvestre,A.J.D.;Freire,C.S.R.;Figueiredo,F.M.L.Nanostructuredbacterialcellulose-poly(4-styrenesulfonicacid)compositemembraneswithhighstoragemodulusandprotonicconductivity.ACS Appl. Mater. Interfaces.2014, 6,7864−7875.. [百度学术]
BagusPambudi,A.;Priyangga,A.;Hartanto,D.;Atmaja,L.Fabricationandcharacterizationofmodifiedmicrocrystallinecellulosemembraneasprotonexchangemembranefordirectmethanolfuelcell.Mater. Today Proc.2020, 46,1855−1859.. [百度学术]
Vilela,C.;Silva,A.C.Q.;Domingues,E.M.;Gonçalves,G.;Martins,M.A.;Figueiredo,F.M.L.;Santos,S.A.O.;Freire,C.S.R.Conductivepolysaccharides-basedproton-exchangemembranesforfuelcellapplications:thecaseofbacterialcelluloseandfucoidan.Carbohydr. Polym.2020, 230,115604.. [百度学术]
Guccini,V.;Carlson,A.;Yu,S.;Lindbergh,G.;Lindström,R.W.;Salazar-Alvarez,G.Highlyprotonconductivemembranesbasedoncarboxylatedcellulosenanofibresandtheirperformanceinprotonexchangemembranefuelcells.J. Mater. Chem. A2019, 7,25032−25039.. [百度学术]
Eikerling,M.;Kornyshev,A.A.Protontransferinasingleporeofapolymerelectrolytemembrane.J. Electroanal. Chem.2001, 502,1−14.. [百度学术]
Vayenas,C.G.;Tsampas,M.N.;Katsaounis,A.FirstprinciplesanalyticalpredictionoftheconductivityofNafionmembranes.Electrochim. Acta2007, 52,2244−2256.. [百度学术]
Zhou,J.;Zhang,R.;Xu,R.;Li,Y.;Tian,W.;Gao,M.;Wang,M.;Li,D.;Liang,X.;Xie,L.;Liang,K.;Chen,P.;Kong,B.Super-assembledhierarchicalcelluloseaerogel-gelatinsolidelectrolyteforimplantableandbiodegradablezincionbattery.Adv. Funct. Mater. 2022 ,32,2111406.. [百度学术]
An,Y.;Yang,Y.;Hu,Z.;Guo,B.;Wang,X.;Yang,X.;Zhang,Q.;Wu,H.High-performancesymmetricsupercapacitorsbasedoncarbonnanosheetsframeworkwithgraphenehydrogelarchitecturederivedfromcelluloseacetate.J. Power Sources2017, 337,45−53.. [百度学术]
Li,H.;Cao,L.;Zhang,H.;Tian,Z.;Zhang,Q.;Yang,F.;Yang,H.;He,S.;Jiang,S.Intertwinedcarbonnetworksderivedfrompolyimide/cellulosecompositeasporouselectrodeforsymmetricalsupercapacitor.J. Colloid Interface Sci.2022, 609,179−187.. [百度学术]
Chen,L.F.;Huang,Z.H.;Liang,H.W.;Guan,Q.F.;Yu,S.H.Bacterial-cellulose-derivedcarbonnanofiber@MnO2andnitrogen-dopedcarbonnanofiberelectrodematerials:Anasymmetricsupercapacitorwithhighenergyandpowerdensity.Adv. Mater.2013, 25,4746−4752.. [百度学术]
Li,K.;Li,P.;Sun,Z.;Shi,J.;Huang,M.;Chen,J.;Liu,S.;Shi,Z.;Wang,H.All-cellulose-basedquasi-solid-statesupercapacitorwithnitrogenandborondual-dopedcarbonelectrodesexhibitinghighenergydensityandexcellentcyclicstability.Green Energy Environ. 2022 ,8,1091−1101.. [百度学术]
Smirnov,M.A.;Tarasova,E.V.;Vorobiov,V.K.;Kasatkin,I.A.;Mikli,V.;Sokolova,M.P.;Bobrova,N.V.;Vassiljeva,V.;Krumme,A.;Yakimanskiy,A.V.Electroconductivefibrousmatpreparedbyelectrospinningofpolyacrylamide- g-polyanilinecopolymersaselectrodematerialforsupercapacitors.J. Mater. Sci.2019, 54,4859−4873.. [百度学术]
Smirnov,M.A.;Vorobiov,V.K.;Kasatkin,I.A.;Vlasova,E.N.;Sokolova,M.P.;Bobrova,N.V.Long-termelectrochemicalstabilityofpolyaniline-andpolypyrrole-basedhydrogels.Chem. Pap.2021, 75,5103−5112.. [百度学术]
Sun,Z.;Thielemans,W.Interconnectedandhighcyclingstabilitypolypyrrolesupercapacitorsusingcellulosenanocrystalsandcommonlyusedinorganicsaltsasdopants.J. Energy Chem.2023, 76,165−174.. [百度学术]
Tanguy,N.R.;Wu,H.;Nair,S.S.;Lian,K.;Yan,N.Lignincellulosenanofibrilsasanelectrochemicallyfunctionalcomponentforhigh-performanceandflexiblesupercapacitorelectrodes.ChemSusChem.2021, 14,1057−1067.. [百度学术]
Sun,Y.;Yang,Y.;Fan,L.;Zheng,W.;Ye,D.;Xu,J.Polypyrrole/SnCl2modifiedbacterialcelluloseelectrodeswithhigharealcapacitanceforflexiblesupercapacitors.Carbohydr. Polym.2022, 292,119679.. [百度学术]
Smirnov,M.A.;Sokolova,M.P.;Geydt,P.;Smirnov,N.N.;Bobrova,N.V.;Toikka,A.M.;Lahderanta,E.Dualdopedelectroactivehydrogelicfibrousmatwithhigharealcapacitance.Mater. Lett.2017, 199,192−195.. [百度学术]
466
浏览量
170
下载量
0
CSCD
相关文章
相关作者
相关机构
